In The News

07 January 2026
Investigator Kamena Kostova, named ‘Cell Scientist to Watch’
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
By Marla Vacek Broadfoot
Proteins are the manifestation of what is written in our genomes—the blueprint of life brought to life. They give structure to cells and tissues, catalyze numerous activities within and between cells, and even control which sections of the DNA blueprint can be read at any given time.
Just as the human genome is the collection of all human genes, the proteome is the sum of all the genome's proteins. But that is where the similarity ends. While the genome locked within every cell remains relatively static, its protein output is constantly changing depending on the cell or tissue, time of day, or exposure to stress. "As a result, there is no single proteome to describe and understand. There are probably an infinite number," says Mike Washburn, PhD, director of the Stowers Proteomics Center.
Despite the daunting complexity of proteomics—or perhaps because of it—Washburn and his colleagues at the Stowers Institute are embracing the field. They believe that studying the proteins of the cell helps lay the foundation of understanding the mechanisms that drive human health and disease.
Tackling the infinite
Proteomics is loosely defined as the study of the structure, function, and interaction of all of the proteins in a particular cell or organism. For human cells, these proteins could number in the hundreds of thousands to over a million. What's more, they could undergo a myriad of modifications, take part in a variety of molecular pathways, or form complicated shapes with multiple protein partners.
The Stowers Proteomics Center is up to the task. Led by Washburn and Laurence Florens, PhD, the Center is staffed by a team of scientists with expertise in protein science, analytical chemistry, biochemistry, computational biology, computer science, and molecular biology. This interdisciplinary design fosters collaboration, which starts with the proteomics team and then radiates out across the Institute and beyond.
"Everything we do is in the context of encouraging people to collaborate with each other and build strong teams around a particular scientific objective," says Washburn. "Our goal is to bring proteomics technologies to bear on important biological problems and stay on top of the technology enough that newer approaches can be introduced to get us there faster."
The proteomics pipeline
When Washburn and Florens, who are married, joined the Institute in 2003, the duo brought with them a powerful proteomics tool—called Multidimensional Protein Identification Technology or MudPIT—that gave Stowers investigators a competitive advantage over their peers. Since then, Washburn, Florens, and the proteomics team have used their technical expertise to adapt other technologies and develop new ones to study the levels of proteins and protein-protein interactions.
Most of these proteomics approaches follow the same basic recipe, with a few modifications. First, the researchers stick a tag on their protein of interest so they can extract it after they test a number of biological conditions. For example, they might want to know how the levels and interactions of a protein change in response to a specific anti-cancer drug. They can grow the protein in cells with and without the drug, and then use the tag to pull out the protein, like a magnet picking a needle out of a haystack. This purification step is particularly complicated, but Charles Banks, PhD, a research specialist in the Washburn Lab, has driven efforts to adopt and improve new purification methods that have become integral to the Proteomics Center and other labs at the Institute.
After purification, the scientists carefully break the proteins into fragments called peptides, turn the resulting concoction from a liquid into a gas, and run it through a high-tech piece of equipment called a mass spectrometer, which identifies the fragments based on mass and charge. These instruments are the cornerstone of proteomics experiments and enable researchers to detect, identify, and quantify tiny amounts of peptides.
In an ongoing effort to refine and improve proteomics strategies, Tim Wen, MS, a senior programmer analyst, and Ying Zhang, PhD, a senior proteomics scientist, have worked closely with Washburn and Florens to overcome technical obstacles and develop new capabilities in the Center. In a series of publications in Analytical Chemistry, they reported a number of new methods for acquiring, processing, and analyzing data, which make it possible to measure the levels of proteins in a sample more accurately and precisely.
Once each of these experimental steps is complete, a challenge still remains—making sense of all the data. Currently, seventy-three terabytes of data reside on the team's hard drive. That amount is roughly equivalent to the volume of information accessible online through the Library of Congress. Fortunately, the Institute has strong information management support to handle this massive amount of data, and the proteomics team includes expert analysts who can sift through data and present it in a way that enables researchers to generate meaningful insights and—in over 170 cases—highly regarded papers.
While Wen writes and maintains software to assemble large datasets rapidly, Gaye Hattem, a programmer analyst, helps researchers determine the biological pathways and functions of the proteins in the datasets, and Mihaela Sardiu, PhD, a senior proteomics scientist, conceptualizes innovative ways to understand and visualize protein interaction networks.
Discovery-driven research
Since its inception, the Proteomics Center has forged many valuable collaborations with other scientists at the Institute. Florens and Anita Saraf, PhD, a senior proteomics scientist, have worked closely with Associate Investigator Kausik Si, PhD, to study the proteomics of learning and memory in fruit flies. Selene Swanson, MA, a senior research specialist in the Proteomics Center, has collaborated frequently with Investigators Joan Conaway, PhD, and Ron Conaway, PhD, to reveal insights about transcription, a key step in the process of constructing proteins from the genetic blueprint. Defective transcription can lead to health problems such as autoimmunity, cardiovascular disease, and cancer. The Conaway Lab has studied many proteins involved in transcription, including thirty individual proteins of a key protein complex called Mediator.
"Without proteomics, this work simply wouldn't be possible," says Joan Conaway. "The Proteomics Center is one of the best things that's happened to us in the last fifteen years."
One of the most frequent visitors to the Proteomics Center is Investigator Jerry Workman, PhD. His research involves histones, the protein spools responsible for wrapping yards of DNA into tight little coils called chromatin. The ability of histones to package DNA is dependent on a number of chemical tags that decorate their surface. The Workman Lab has identified several groups of proteins that are responsible for modifying histones, either by attaching or removing these chemical tags or through some other means, and many of these proteins have been implicated in cancer.
A basic scientist at heart, Workman was interested in histones and chromatin remodeling long before their links to cancer brought them into the spotlight. But now, he uses proteomics technologies to see how these complexes are working in the cell, in the context of both health and disease.
"Proteomics is all about the power of discovery," says Workman. "There's a feeling in the life sciences that we have to focus on hypothesis-driven research, but with the Stowers Proteomics Center, we can do discovery-driven research. We might have an interesting protein that we think must be interacting with other proteins that are interesting. We don't know what they are, so we don't have a hypothesis, but we can use proteomics to find out. And more often than not, we will uncover interactions that we never could have predicted."
Recently, Workman was surprised to find a series of metabolic enzymes that are also involved in chromatin remodeling. One of these proteins, called pyruvate kinase M2 or PKM2, is overexpressed in human tumors and has been shown to reprogram metabolism in cancer cells. Workman and his colleagues purified the yeast version of the protein and used it to identify a handful of other metabolic enzymes that regulate histone modifications. They plan to use proteomics to characterize each of those proteins and determine how they manage to pull off their dual roles.
Workman's collaboration with the Proteomics Center has paid off in more ways than one. Their joint projects have produced over three dozen publications. One of their most successful projects formed the basis of a National Institutes of Health grant proposal, which was recently funded and will support a new line of research in Washburn's laboratory.
A broader impact
It all started with a conversation between Karen Smith, PhD, a former postdoc in the Workman Lab, and Sardiu in the Proteomics Center. Smith was trying to identify the members of Sin3/HDAC, a protein complex known to pop the acetyl groups off histones in order to silence genes. Histone deacetylases, or HDACs, often repress genes that would normally keep cells from growing out of control, and have become popular targets for cancer therapy. Sardiu, who was helping Smith take her protein inventory, suggested they take the project even further. She devised a way to determine statistically not only what was in the complex, but also how the entire complex was put together.
The researchers used the method to analyze Sin3/ HDAC, both alone and in the presence of a histone deacetylase inhibitor called SAHA. This inhibitor is currently approved for treatment of T-cell lymphoma and is being investigated in clinical trials for use in many other types of cancer, but its mode of action remains a mystery. Smith and Sardiu found that SAHA doesn't simply quench the activity of the histone deacetylase, but actually changes the architecture of the entire complex. Their results were published last year in the journal Molecular & Cellular Proteomics.
Washburn was recently awarded a five-year, $1.5 million grant from the NIH to expand the project. Together with Banks and Janet Thornton, a senior research technician, he will use proteomics technologies, as well as biochemical, computational, and genomic techniques, to tease apart further the mechanism of action of SAHA. In the end, Washburn hopes the research will yield a better understanding of how the inhibitor works, and perhaps even lead to the development of new HDAC inhibitors.
"We are in a position to ask and answer some questions about this cancer treatment that probably not a lot of people can," says Washburn. "Cancer is such a devastating illness, and seems to affect everyone at one point or another. If we can make a difference, it is certainly worthwhile."
In addition to working with nearly all the laboratories at the Stowers Institute, the Center has forged over a dozen collaborations with scientists around the world. One external collaboration focuses on decoding the malaria histone code and another involves uncovering interactions between the human immunodeficiency virus (HIV) and human immune cells. Both have generated insights into these persistent and deadly infections.
Concerted team efforts are key to advancing the complicated field of proteomics. Through multidisciplinary expertise and teamwork, scientists are gaining a better glimpse into the complicated inner lives of our cells and the proteins that make us tick.
"Proteomics enables us to ask basic questions about how things work and how things interact," says Washburn. "The answers this research generates could be applied to critical human health issues with a level of precision and detail that was never before possible."
Proteomics research generates large amounts of data — how do scientists make sense of it? One way is by using network maps to visualize protein connections based on their physical interactions. Each node in the network represents an individual protein and interacting proteins are linked. Network maps can also show how protein interactions change over time or under different conditions.
The image above shows part of a protein network map from a study involving SAHA, a molecule used to treat certain cancers. The researchers analyzed protein interactions in cells from a human cell line before and after treatment with SAHA. Green links correspond to interactions that increased in abundance after SAHA treatment while magenta links represent interactions that decreased. The thickness of the lines corresponds to the degree of the effect. Structures are shown for some of the proteins in the network map.
How SAHA works against cancer is not completely understood, but studies like this help scientists glimpse changes in the protein network upon treatment. Researchers can get an idea of how widespread a molecule's effects are and may identify proteins in the network to investigate further.
In The News

07 January 2026
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
#Stowers25: Celebrating 25 Years
06 January 2026
Alejandro Sánchez Alvarado, Ph.D., reflects on a year of discovery, gratitude, and the community that helps support our mission.
Read Article
In The News
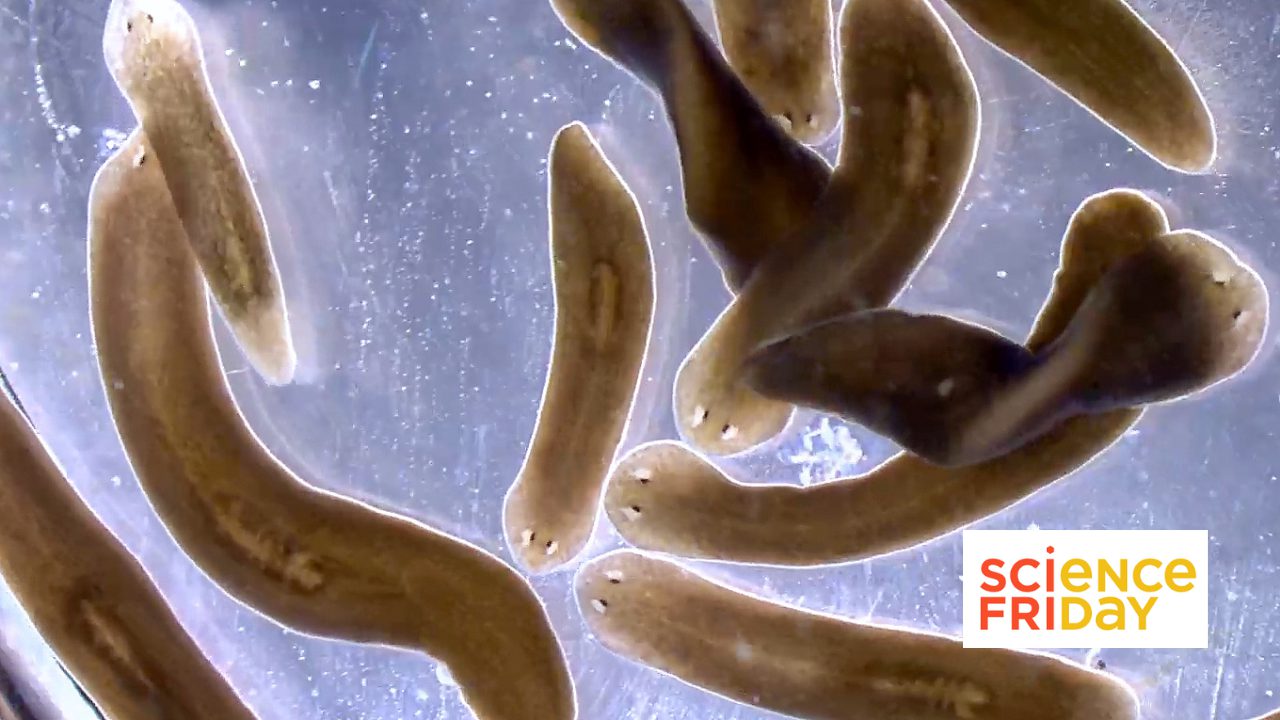
01 January 2026
From Science Friday, President and CSO Alejandro Sánchez Alvarado talks about the science of regeneration and the biology lessons we can carry into the new year.
Read Article
