Organisms
Investigating research organisms to shed light on human health
From traditional model research organisms like fruit flies to unusual organisms like sea anemones, research organisms at the Stowers Institute are carefully chosen to answer a question or help solve a biological mystery. Explore some of the organisms we study below.
(Un)ordinary Organisms
Since the very first cell emerged, life on Earth has evolved in astounding ways. Given the immense diversity of different forms of life, many of the genetic and cellular processes have both evolved and been conserved. Research organisms from single-cell yeasts and zebrafish to chameleons and mice can provide clues to the processes governing human health and disease. Learn more about these organisms found all around us, capable of unordinary biological feats.
Learn more about our (un)ordinary organismsDiscovery made possible through nature
"When I see these organisms, I see untapped potential."
Apple snails

Apple snails
Apple snails possess an eye very similar to a human eye that can completely regenerate.
Apple snails are a diverse species of freshwater mollusk found on nearly every continent. While in some cultures, they are considered a culinary delicacy, they are invasive, and voracious, animals that continue to wreak havoc on agricultural and natural wetland ecosystems, after either intentional or accidental introduction. However, they are a unique and valuable research organism for studying evolution and regeneration.
Why
At the Stowers Institute, sealed tightly shut in a lab with extensive precautions taken as to not accidentally introduce the invasive species to the Kansas or Missouri water systems, over 1000 apple snails of species, Pomacea canaliculata, are housed and studied. Due to their long evolutionary history and expansive diversity in morphology, physiology, and geographic distribution, these mollusks are an excellent research organism for studying evolution and comparative genomics. In addition, the eye of the apple snail closely resembles the vertebrate eye. When removed, the snail’s eye regenerates, providing insight into the mechanisms governing eye regeneration of the closest approximation to a human eye found so far.
Applications
How the apple snail eye regenerates post-amputation has implications for uncovering the basic mechanisms underpinning regeneration across different species. In humans, damage and disease can lead to irreversible ocular loss of function, or blindness. Remarkably, the eye of the apple snail closely resembles vertebrate eyes. Understanding the ability of apple snail cells to regenerate all eye components—cornea, retina, and lens—in adults and the degree of overlap between this process and embryonic eye development may lead to treatments applicable in humans.
Learn more about our apple snail research here.
Labs studying apple snails
Sea lamprey

Sea lamprey
The sea lamprey, a carnivorous vertebrate lacking a jaw, is an important research organism for studying animal evolution.
Native to the Atlantic Ocean, the sea lamprey, Petromyzon marinus, sits at the base of the vertebrate tree of life. Lacking a jaw, these vertebrates split from jawed vertebrates over 500 million years ago. The sea lamprey has unusual traits like the ability to regenerate its spinal cord and is extraordinarily resilient—it has survived four global extinction events while maintaining its morphology and physiology, for at least 300 million years.
Why
The sea lamprey gained notoriety in the mid-20th century when these eel-like creatures invaded the Great Lakes in the U.S. and Canada, wreaking havoc on fisheries. Collaborations between government and research institutions have largely mitigated the sea lamprey’s destruction by reducing its population. However, as a research organism, scientists are increasingly using the sea lamprey to investigate evolutionary biology as it pertains to vertebrate diversification and complexity, development, regeneration, and the immune system. Stowers scientists are looking at the sea lamprey for insights into the gene regulatory networks governing key events during early development.
Applications
Due to their unique position on the vertebrate tree of life, investigating the sea lamprey can provide powerful insights into how animals,’ including humans,’ complexity have evolved. Some processes unique to vertebrates like hindbrain development are proving to be exceptionally conserved between jawed and jawless species, which have important implications for understanding our own evolutionary development. Additional applications for studying the sea lamprey involve understanding the mechanisms that allow them to regenerate their spinal cord and how they have developed a unique adaptive immune system.
Learn more about our sea lamprey research here.
Labs studying the sea lamprey
Planarians

Planarians
Planarians are wildly diverse flatworms renowned for their astounding regenerative capabilities
Geographically located in almost every corner of the globe, planarians are free-living flatworms that can be found in fresh water, marine environments, and even on land. Hundreds if not thousands of unique strains and species exist and different types can either reproduce asexually and/or sexually.
Why
Planarian research organisms are an exceptional system to uncover cellular and molecular mechanisms underlying regeneration, the replacement of differentiated cells from stem cells following injury or natural physiological turnover. Although certain animals including humans have regenerative capabilities in many tissues and organs, we have limited or no ability to replace missing limbs, or to repair damage to the heart or brain. Insights into the mechanisms governing regeneration are also essential for understanding normal development in higher organisms including humans.
Applications
Investigating the remarkable ability of planarian stem cells to differentiate into any and all cells needed for organism regeneration enables identification and characterization of the molecular mechanisms involved. These processes have important and practical applications for understanding normal development, and perhaps to engineer similar mechanistic programs to eventually bypass the need for organ transplantation.
Learn more about our planarian research here.
Labs studying planarians
Fruit flies

Fruit flies
Despite their reputation as a nuisance in the kitchen, fruit flies have been a valuable mainstay for biological research for more than 100 years.
The long history of research on fruit flies—and specifically the species Drosophila melanogaster—has generated a deep knowledge base and extensive set of tools and techniques for this research organism. Although they don’t have backbones and look quite dissimilar to humans, their genomes and other fundamental aspects of biology are surprisingly similar.
Why
Fruit fly research has uncovered many key insights into biology across disciplines including genetics, development, neuroscience, evolutionary biology, and behavior. Easily cultured in the lab and able to produce many offspring in a relatively short generation time, fruit flies offer a powerful system for discovering how life works, from the principles of inheritance to how cells, tissues, and organs develop and function.
Applications
The emergence of gene and genome sequencing technologies over the past two decades has created an explosion of genomic data available for fruit flies, humans, and many other species. About 75% of known human disease genes have an identifiable match in the fruit fly genome. Fruit fly research has increased our understanding of molecules and mechanisms that become compromised in human health conditions and has uncovered drug targets for treating these conditions. The tractable complexity of fruit flies will continue to allow scientists to explore developmental, behavioral, and evolutionary questions, and relate this insight to other research organisms and humans.
Labs studying fruit flies
Mice
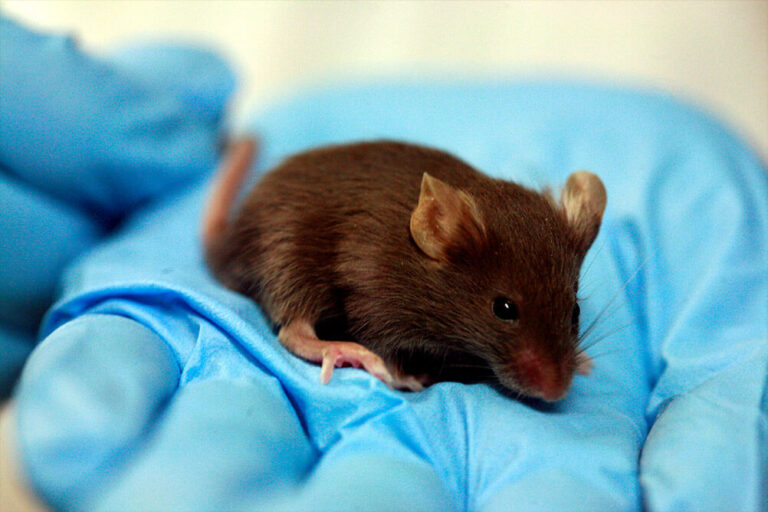
Mice
Mice are the most common research organism used for studying human disease in the world.
As fellow mammals, mice are very similar genetically and physiologically to humans. Almost all genes in mice have counterparts to genes in humans. This genetic similarity gives rise to many other parallels, such as how we develop from a fertilized egg to an adult and the collection of tissues and organs in our bodies. Our various biological systems – reproductive, digestive, hormonal, and nervous, to name a few – are also very similar to those of mice.
Why
Mouse research enables a broad range of investigation of genes and mechanisms responsible for aspects of development and normal adult biological function, including chromosome integrity, regulation of gene activity, cell division, cell fate and migration, sensory perception, behavioral responses, and aging. The ability to alter the mouse genome using genetic and gene-editing approaches, the ease of inbreeding to generate genetically identical strains, their accelerated lifespan, and decades of prior studies contribute to their prevalence in biological research.
Applications
In addition to genetic, developmental, and physiological similarities to humans, mice are also afflicted with many of the same diseases and health conditions such as cancer, hypertension, diabetes, and osteoporosis. Mouse research at Stowers is providing answers to a wide range of biological investigations in areas such as craniofacial and other congenital conditions, body patterning, metabolism, adult stem cell regulation, odor processing, and memory formation.
Labs studying mice
Sea anemones

Sea anemones
The sea anemone is a marine animal used to study toxin delivery, regeneration, and aging.
The sea anemone, Nematostella vectensis, is a species of cnidarian, a type of water-dwelling sea creature ranging from corals to jellyfish. With an evolutionary history dating back between 500-700 million years, these animals share similar stinging structures used for predation and protection.
Why
Nematostella are excellent research organisms for investigating regeneration, the lack of senescence—the decline in cellular function over time—and their unusual and complex stinging mechanism used to capture prey and to protect themselves from predators. Although sea anemones have been the subject of scientific study for over a century, recent advances in technology have enabled characterization, particularly of their stinging organelles, in spectacular detail.
Applications
The structure and mechanism of the cnidarian stinging organelle has incredible applications for designing targeted medical delivery devices for employment at microscopic levels. Additional applications may involve uncovering the mechanisms that underlie Nematostella immortality. Scientists may be able to learn from this cnidarian how to mitigate premature cellular and organ decline in humans.
Labs studying sea anemones
Yeast
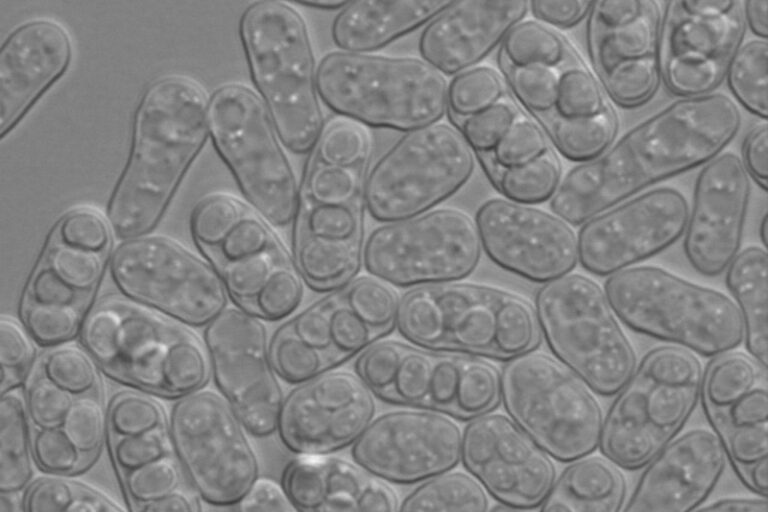
Yeast
With over 1000 unique yeast species classified, these single-celled eukaryotes are a widely studied microorganism belonging to the fungus kingdom.
The vast variety of yeast species fall into two primary categories: fission and budding yeast. Budding yeast strains have been utilized by humans for thousands of years in culinary and fermented beverage preparation. Fission yeast strains along with budding yeast are exceptional research organisms for studying genetics and reproduction.
Why
The genetic tractability and ease of controlling reproduction cycles in yeast have established these single-celled organisms as an invaluable system for studying genetics and cell life cycles.
With an evolutionary history dating back hundreds of millions of years, different yeast species are staples in research laboratories as well as in baking and brewing. Scientists at the Stowers Institute study and maintain a large variety of natural and lab isolates of both fission and budding yeast, primarily Schizosaccharomyces pombe and Saccharomyces cerevisiae, respectively. Both types of yeast primarily reproduce asexually, where smaller cells are formed from a mature budding yeast by “budding,” while fission yeast reproduce mitotically through fission, or dividing down the cell’s center to create two identical cells. However, in the lab and in nature, both types can mate and undergo sexual reproduction, typically in nutrient-stressed conditions. In the lab, mating and meiosis can easily be induced to investigate the stages of sexual reproduction and genetic inheritance.
Applications
Due to the relative simplicity of their genome, yeasts are some of the most common and powerful research organisms used in the study of genetics and cell biology. The ability to manipulate genes allows investigation of gene expression, transcription and translation, and protein expression and dynamics. In addition, since inducing meiosis is extremely easy, labs at the Institute use yeast to investigate sexual reproduction. The applications for controlling the timing of meiosis have led to breakthroughs in understanding female reproductive aging and the dynamics of how selfish genes can bias transmission of genetic information to offspring, typically at the expense of organism fitness and fertility. These microorganisms have tremendous potential for understanding infertility, aging, and inflammatory diseases in humans.
Zebrafish

Zebrafish
Zebrafish are freshwater vertebrate animals used to study development, genetics, and regeneration.
The freshwater zebrafish native to South Asia is characterized by lateral line sensory systems. They contain sensory organs called neuromasts dotted along lines on the head and sides of their bodies. Stowers has an extensive collection of zebrafish to accommodate the six labs currently conducting research on these organisms.
Why
Zebrafish are powerful research organisms for investigating a variety of topics. Their embryonic development is extremely rapid and zebrafish larva are transparent which enables visualization of developmental processes. At the embryonic stage, zebrafish heart, lateral line sensory organs and fins regenerate quickly and have enabled Stowers researchers to characterize molecular, cellular, and transcriptional processes at very high spatial and temporal resolution. These mechanisms serve as platforms for studying similar processes in different organs and species.
Applications
The many applications of zebrafish research include the mechanisms involved in development, regeneration, genetics, neural crest cells, cancer, and reproduction. For example, the Piotrowski Lab has used the zebrafish lateral line sensory hair cells, named such as they closely resemble the hair cells in the human inner ear, to identify processes involved in repair and regeneration of these cells to address hearing loss in humans. The Trainor lab uses these animals to study neural crest development that can be applied to congenital defects like Treacher-Collins Syndrome.
Labs studying zebrafish
Killifish

Killifish
Killifish are freshwater vertebrates used to study regeneration, aging, and diapause, a state of suspended development in animals, allowing them to survive extreme environmental conditions.
Killifish, a diverse group of over 1,000 freshwater species of fish, inhabit lakes, streams, and ponds on nearly every continent. They are generally fairly small, ranging in size from 1 to 3 inches, with some growing as large as 6 inches. Their short lifespans, robust regenerative capabilities, and ability to pause development in response to harsh or extreme environmental conditions make them valuable systems for studying regeneration, aging, and diapause.
Why
Some killifish species live in flooded pools of water that periodically dry up depending on the season. Known as annual killifish, these fish are ideal systems for studying aging and age-associated conditions including neurodegeneration and cancer due to their remarkably short lifespans (three to nine months). The African turquoise killifish, Nothobranchius furzeri, is the shortest-lived vertebrate species in laboratory environments and notably, lacks genetic markers commonly associated with aging. In addition, after fertilization and initial body plan formation, annual killifish can suspend their growth, for months or even years if environmental conditions are unfavorable, before resuming normal development. Many species of killifish also exhibit regeneration, including some regrowing their fins, a surprising adaptation given their short lifespan.
Applications
Killifish biology offers valuable insights into fundamental biological processes. At the Stowers Institute, scientists are exploring the cellular and molecular mechanisms behind killifish regeneration. Recently, three new killifish species from Africa and South America arrived at the Institute to help Stowers scientists uncover why some species undergo embryonic diapause while closely related species do not. These remarkable features make killifish powerful research organisms for advancing our understanding of aging, regeneration, and developmental biology.
Labs studying killifish
Coral

Coral
Coral are marine invertebrates that can form large colonies, serving as underwater ecosystems supporting 25% of ocean biodiversity.
Closely related to sea anemones and jellyfish, around 6,000 species of coral live in oceans around the globe, with most inhabiting tropical shallow waters and some polar species residing at great depths. Coral, which belong to a group of animals called cnidarians, possess stinging organelles to capture prey. However, more tropical coral receive most of their energy and nutrients via symbiosis from algae they house. Algae converts sunlight into food through photosynthesis and lends coral their unique colors. Many of these marine invertebrates, whose origins can be traced to nearly 600 million years ago, are critically endangered due to rising sea levels and temperatures.
Why
Coral are studied to gain insights into evolutionary origins of complex animals, as well as the biological process of symbiosis. Coral are capable of both sexual—the formation of new individuals called polyps—and asexual reproduction—where polyps reproduce to form interconnected colonies of coral. Pollution and sea temperature increases due to climate change can result in coral bleaching events when their symbiont algae are expelled, and often coral death. For reef-building coral, which both harbor a quarter of marine life and buffer coastal regions from storms, the endangerment of this vital animal is threatening coastal populations’ livelihood and property.
Applications
Climate-induced rises in sea level and temperature along with ocean acidification—the ocean absorbs roughly 90% of atmospheric carbon dioxide—are causing mass coral reef bleaching and death. Given the essential role of coral for sheltering marine biodiversity and their critical endangerment, Stowers scientists are investigating coral biology from both an evolutionary as well as a conservational perspective. Stowers Institute Investigator Matt Gibson, Ph.D., is building upon his lab’s research with the sea anemone, Nematostella, a cousin of coral, to gain further insight into the biology of cnidarians and to potentially engineer more robust coral that can survive in a changing climate.
Labs studying coral
Sea squirt

Sea squirt
The sea squirt, a common name for tunicates, is an ancient marine invertebrate used to study evolutionary and development biology.
Tunicates, also known as sea squirts or sea tulips, are a diverse group of more than 3,000 marine species inhabiting shallow ocean waters around the world. While fossil records indicate that these invertebrates emerged over 500 million years ago, they share genetic similarities to vertebrates. During early development, tunicates possess a notochord—a rod-like structure which, in more complex animals, eventually develops into a backbone. This makes the tunicate important for studying the evolution of chordates, a broad class of animals including humans.
Why
Studying the biology of tunicates, ancient invertebrates with surprising similarities to vertebrates, can help us understand how animals like fish, birds and mammals evolved. As larva, tunicates have a simple backbone-like structure called a notochord that disappears as they mature. In more complex animals, including humans, this develops into the spine during early growth. Because of their shared genetic traits, tunicates are classified as chordates, a group that includes all animals that can form precursors to backbones. Tunicates develop quickly, have a relatively simple body plan, and are transparent during development, allowing scientists to track cellular specialization and gene expression.
Applications
Research on tunicates has applications toward a greater understanding of evolutionary and developmental biology for animals spanning the tree of life. The lab of Investigator Tatjana Sauka-Spengler investigates neural crest cells and the gene regulatory networks, or circuitry, that guide them across a broad swath of species. While neural crest cells, the embryonic stem cells that form many parts of the adult body, are unique to vertebrates, tunicates have neural crest-like cells. Stowers scientists in the Sauka-Spengler Lab are using tunicates to try to build similar networks in an invertebrate chordate system, which may yield new knowledge underlying the genetic pathways governing vertebrate development.
Labs studying sea squirts
Alfalfa

Alfalfa
Alfalfa is a flowering legume that serves as a valuable organism for studying how plants and bacteria work together. These interactions allow plants to utilize nitrogen from the air instead of depending on nitrogen fertilizers from soil.
Alfalfa, a flowering legume native to temperate regions including North America, Europe, South Africa, and Australia, has been cultivated to feed livestock for thousands of years. Legumes like alfalfa are used to study symbiosis—a mutually beneficial relationship between two unrelated species, in this case, plants and bacteria. This symbiosis enables bacteria to convert inorganic nitrogen found in the atmosphere to a usable form needed for plant life.
Why
Symbiosis is a powerful system where two unrelated species live together to benefit each other. Common to nearly all life forms, including humans, the mechanisms underlying this complex process can be better understood by studying the simpler, one-to-one relationship alfalfa forms with the nitrogen-converting bacteria, Rhizobium. In addition, alfalfa is easy to grow and has a relatively simple genome, which is helpful for dissecting the molecular and genetic bases underlying plant-microbe symbiosis, with potential agricultural and human health applications.
Applications
Researching alfalfa has broad applications for plant biology and agriculture. Many plants, including alfalfa, produce small protein-like molecules called peptides that help control how plants and bacteria work together to retrieve nitrogen from air. The lab of Stowers Institute Assistant Investigator Siva Sankari, Ph.D., is researching how certain peptides have evolved to precisely control bacterial metabolism, allowing the plant to fine-tune this relationship for optimal growth. The Sankari Lab also investigates how plants and bacteria manage essential trace metals like iron and zinc, which must be carefully balanced within the symbiotic partnership. These discoveries are helping develop new approaches to improve crop performance, reduce reliance on fertilizers, and explore medical applications inspired by how these peptides influence microbes.
Labs studying alfalfa
(un)ordinary organisms
We have more in common than you may think
Learn about our Labs

Our diverse labs are united in their commitment to fostering an innovative and collaborative environment in the pursuit of foundational biomedical knowledge.
