In The News

07 January 2026
Investigator Kamena Kostova, named ‘Cell Scientist to Watch’
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
News
New methods on planarian flatworm stem cells mark first step toward establishing transgenic planaria

Transplanting pluripotent stem cells into a host planarian flatworm during the Planarian Transgenesis Workshop.
By Rachel Scanza, Ph.D.
GloFish, the vibrant, glow-in-the-dark tetra fishes you may have seen in pet stores are products of transgenics. Transgenesis is the process of transferring genetic information from one organism into another’s genome. For the fish mentioned above, genes encoding fluorescent proteins are inserted into the genome of normal fish, making them literally glow.
From a scientific perspective, investigating and perfecting this research technique within the whole-body regenerating flatworm, Schmidtea mediterranea, could provide scientists with powerful tools to understand regenerative processes at unprecedented resolution. Uncovering primary principles at the molecular and cellular level may allow the development of approaches to induce regeneration in other organisms, including humans.
The Stowers Institute for Medical Research hosted the Planarian Transgenesis Workshop, funded by the National Science Foundation EDGE program in November 2022. Scientists from around the world studying flatworms—from India to Spain to Israel—learned how to insert a reporter gene—a nucleic acid sequence that can “report” or indicate that the transplantation method is viable—into a live worm and observed cells generating the protein in real time.

Image of host planarian flatworm rescued by transplanted stem cells (dpt: days post transplant).
“The recent workshop is an example of best practices in a scientific community—sharing key methods, reagents, and technologies. Twenty institutions spanning three continents participated in this collective experience,” said Stowers Executive Director and Chief Scientific Officer Alejandro Sánchez Alvarado, Ph.D.
The workshop was conceived in part from a new study started by Kai Lei, Ph.D., when he was a postdoctoral researcher in the Sánchez Alvarado Lab and completed in Lei’s independent laboratory at Westlake University. The researchers established a novel, reliable method to keep planarian stem cells alive in an artificial environment, or cell culture, and showed that the cultured cells could be transplanted into flatworms devoid of stem cells and rescue their viability. Published in iScience on February 17, 2023, this technique was able to rescue planarians after applying a lethal dose of radiation. In addition, this work represents a new way to observe the delivery and expression of RNA, and potentially DNA, in a host planarian flatworm post-transplantation.
“Planarian researchers have been eagerly trying to culture stem cells for the past two decades and to manipulate them genetically,” said Postdoctoral Researcher Anand Sasidharan, Ph.D., from the Sánchez Alvarado Lab. “Recent efforts from a fruitful, multi-institutional collaboration along with the Planarian Transgenesis Workshop is beginning to pave the way for genetically modifying the planarian genome.
Pluripotent stem cells dividing in culture.
Pluripotency refers to the ability of a cell to differentiate, or turn into, any type of cell in the body. This naturally occurs early in development when the very first cells must decide their fate—do I become a muscle cell or a brain cell? Planarians are a widely studied organism because they maintain pluripotent cells into adulthood and possess a subpopulation of cells that can regenerate an entirely new worm.
The team extracted these cells and developed a culture medium for them to reside and retain their pluripotency, outside of their host. In the new culture, stem cells could complete cell division and be transferred by injection into a different flatworm and continue to divide. In fact, after transplanting stem cells from an asexually reproductive strain of the planarian flatworm S. mediterranea into an irradiated sexually reproductive strain, the worm changed its procreative preference.
“This a first step towards culturing and manipulating planarian pluripotent stem cells,” said Sánchez Alvarado. “The methods described in our report should significantly help the community in its collective effort to generate transgenic organisms as well as to help introduce changes to the planarian genome.”

Graphical abstract illustrating the cycle of extracting pluripotent stem cells, allowing them to proliferate in culture, and transplanting them into irradiated flatworms. Bottom right illustrates the expression of mRNA in a host cell.
An additional objective was to optimize injecting a gene or its transcribed form, messenger RNA (mRNA), into a host’s particular genome and to observe its incorporation. The beauty of this research? If we can change DNA or RNA in stem cells pre-transplantation, we can generate transgenic planarians to investigate and possibly uncover first principles of pluripotency, and thus regeneration at the molecular level.
“By broadly and openly disseminating this new ability to introduce a reporter gene into planarian cells, our chances to permanently modify the planarian genome to better understand animal regeneration are tangibly increased,” said Sánchez Alvarado.
This study marks the first report of pluripotent stem cells completing cell division in vitro, or outside an organism. And when injected into a planarian flatworm relieved of its stem cells, the newly transplanted cells proliferated and spread throughout the worm, rescuing it from imminent demise. The potential for understanding, and perhaps inducing regeneration in organisms higher up the phylogenetic tree is becoming a possibility instead of a pipe dream.
Additional authors include Wenya Zhang, Jiajia Chen, Ph.D., Sean McKinney, Ph.D., Eric J Ross, and Heng-Chi Lee, Ph.D.
This work was funded by the National Institutes of Health (NIH) (award: R37GM057260) and the National Natural Science Foundation of China (award: 31970750). The workshop was funded by the National Science Foundation EDGE program. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
In The News

07 January 2026
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
#Stowers25: Celebrating 25 Years
06 January 2026
Alejandro Sánchez Alvarado, Ph.D., reflects on a year of discovery, gratitude, and the community that helps support our mission.
Read Article
In The News
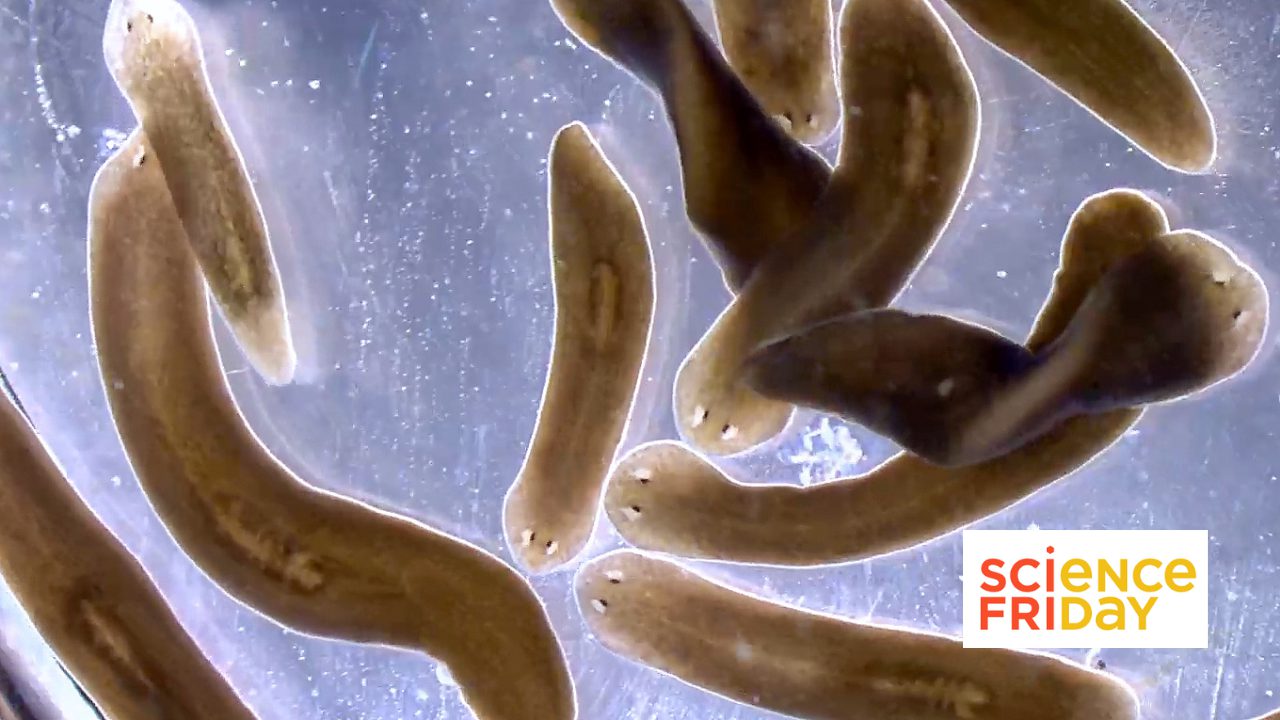
01 January 2026
From Science Friday, President and CSO Alejandro Sánchez Alvarado talks about the science of regeneration and the biology lessons we can carry into the new year.
Read Article
