In The News

27 May 2026
Amputated sea cucumber tissue keeps living for years—possibly forever
From Scientific American, President Alejandro Sánchez Alvarado, Ph.D., shares expert insight on a fascinating new regeneration study.
Read Article
Press Release
Stowers scientists expand their pivotal discovery on the form and function of amyloids
KANSAS CITY, MO — January 26, 2026 — Why some memories persist while others vanish has fascinated scientists for more than a century. Now, new research from the Stowers Institute has identified the mechanism that makes a fleeting moment unforgettable. In a study that culminates more than 20 years of work, Stowers scientists have provided the first direct evidence that the nervous system can deliberately form amyloids to help turn sensory experiences into lasting memories. The research forces us to rethink long-standing assumptions about memory and the consequence of amyloid formation in the brain, potentially providing new avenues for treating amyloid-related disorders of the nervous system.
“I wanted to understand how unstable proteins help create stable memories,” said Stowers Institute Scientific Director Kausik Si, Ph.D. “And now, we have definitive evidence that there are processes within the nervous system that can take a protein and make it form an amyloid at a very specific time, in a specific place, and in response to a specific experience."

Kausik Si, Ph.D.
Set to publish in the Proceedings of the National Academy of Sciences on January 30, 2026, the study from the Si Lab focuses on "chaperone proteins" in the fruit fly. Chaperones guide other proteins to attain the correct folded state. In the nervous system, they have long been thought to either help proteins fold correctly or prevent harmful misfolding and clumping. But the researchers discovered a specific type of chaperone that does something unexpected — it allows proteins to change their shape and form functional amyloids that house long-term memory.
“This expands the idea of a protein’s capacity to do meaningful things, and suggests there is an unknown universe of chaperone biology that we’ve long been missing,” Si said.
Amyloids are typically associated with neurodegenerative diseases such as Alzheimer’s, Huntington’s and Parkinsons. They form tightly packed, highly stable “detrimental” protein fibers that destroy brain cells, erasing the memories of their host.
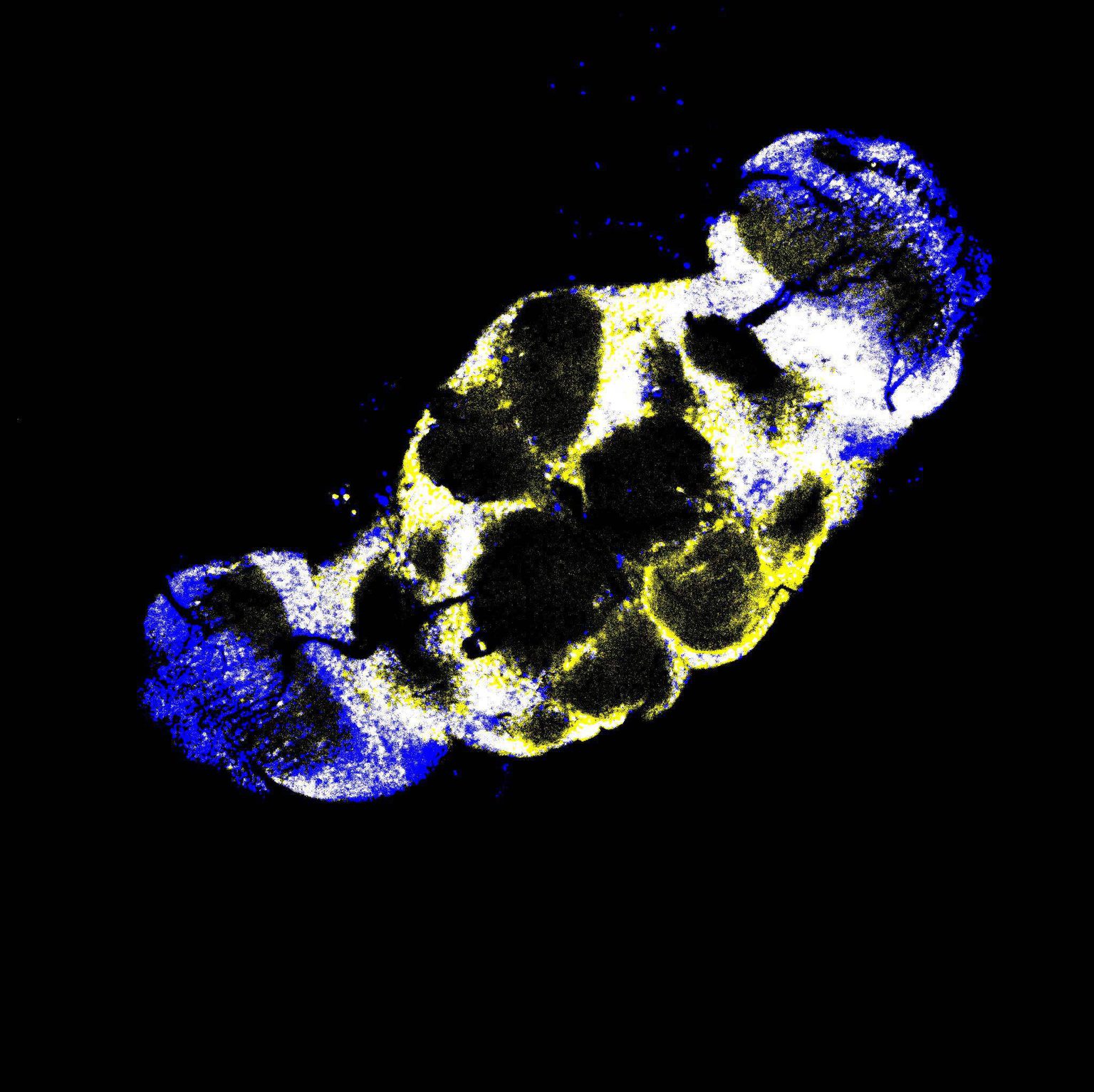
Fluorescent microscopy image of the fruit fly brain stained for cell nuclei (blue) and Funes (yellow).
The new research not only strengthens the Si Lab’s paradigm-shifting 2020 Science study which explained amyloids are not always harmful, unregulated byproducts as previously thought; it also proves amyloids can be carefully controlled — serving as tools the brain uses to store information. Ultimately, the research reveals for the first time a critical step in the process of how long-lasting memories endure.
"Discovering this chaperone protein has now provided us with an avenue to potentially approach amyloid-based diseases in an unanticipated way,” Si said. “It may be possible to either activate these chaperones and guide toxic amyloids to be less harmful — or, by activating them, we can potentially endow the brain with enhanced capacity to form functional amyloids. This could then override the intrusion of disease-causing amyloids.”
The Si Lab knew from their 2020 study, led by former Stowers Postdoctoral Research Associate Rubén Hervas, Ph.D., now an Professor at the University of Hong Kong and co-corresponding author on the current study, that the formation of amyloids can allow animals to form stable memories.

Rubén Hervas, Ph.D.
"But we did not know how or when,” Si said.
“The fact that amyloid is needed to form memory implied there must be a mechanism that controls the process,” Hervas said. The researchers hoped that if they could identify the mechanism, they could also manipulate it in a meaningful manner to influence memory.
“Despite 100 years of studying amyloid biology, nobody has ever asked how the brain can deploy amyloid,” Si explained. “Because amyloid formation was historically thought to be unintentional and unintended, it was now necessary for us to ask that question.”
For more than a century, amyloids have been considered harmful and were assumed to arise when the nervous system fails to control misfolded proteins. The new study rethinks that assumption: all proteins linked to amyloid, including those implicated in disease, are expressed throughout the body from birth, yet amyloid forms only in the nervous system and often late in life. Si weighs two possibilities — a slow, gradual accumulation, or a purposeful deployment for a normal physiological role that can become harmful over time. He argues the latter is more likely.
In 2003, Si first discovered the existence of a functional amyloid in the sea slug. With just 10,000 neurons, this simplified system was a first but pivotal step toward rethinking amyloid biology. His lab then expanded the research to more complex animals including fruit flies (~150,000 neurons), mice (70-80 million neurons), and even humans (~86 billion neurons). The team eventually uncovered that an amyloid-based mechanism is broadly used for memory persistence.
In fruit flies, a prion-like protein called Orb2 (and its relative protein CPEB in mammals) must undergo self-assembly at the synapses, the gap between two neurons, to maintain a memory. Over time, the researchers began to hypothesize that the difference between a harmful and a helpful amyloid may depend on whether Orb2’s assembly process is tightly regulated by other proteins.
This new study allowed the team to finally test that hypothesis. To find the regulator, they investigated a family of chaperones that manage protein behavior in neurons and, using an associative memory model, they identified a previously uncharacterized chaperone.
“We were inspired by Jorge Luis Borges’ short story Funes the Memorious in which one man’s perfect memory comes at a cost, so we named the chaperone Funes,” said Kyle Patton, Ph.D., a former Stowers Graduate School student and lead author on the study.
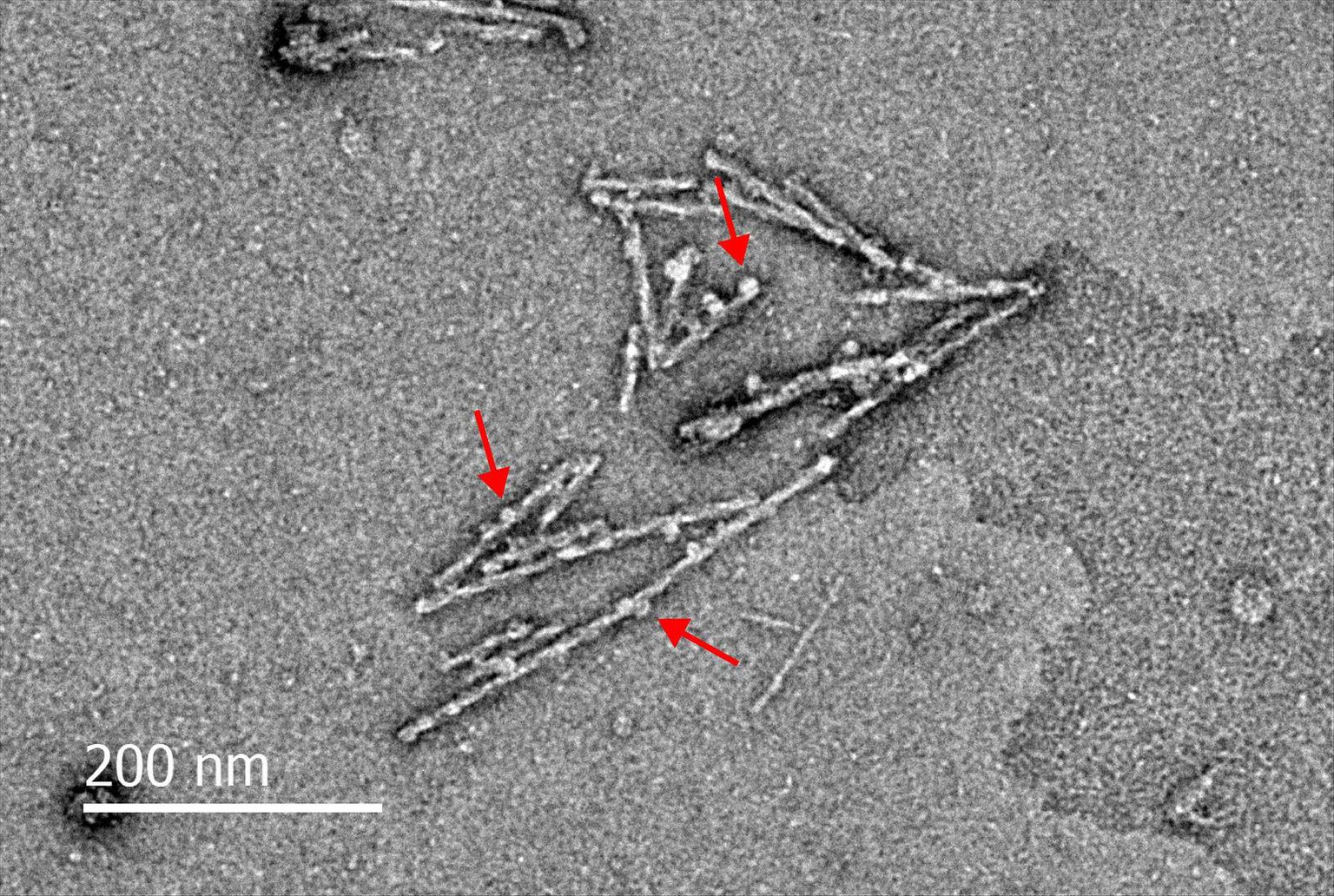
Electron microscopy image of Orb2 amyloid fibers. Red arrows indicate the chaperone protein Funes aiding in the assembly of Orb2 amyloid.
The researchers discovered Funes by manipulating the concentrations of 30 different chaperones in the fly’s memory centers.
“We trained very hungry fruit flies to link a specific, unpleasant smell with a sugar reward,” Patton said. Flies with increased levels of Funes showed a remarkable ability to remember the odor-reward link after 24 hours — a standard proxy for long-term memory.
But the most surprising discovery came at the molecular level. At his lab in Hong Kong, Hervas engineered Funes variants that could bind Orb2 but could not trigger its transition into amyloid, and found the flies’ long-term memory failed. This indicated that Funes is an essential component for long-term memory formation.
“We are now getting early evidence that, like the fruit fly shows in this study, the process may also be manifesting in the vertebrate nervous system,” Si said. “Our hypothesis is carrying us all the way to the vertebrate brain, illustrating that it may actually be universal.”
Discovered nearly 50 years ago, chaperone proteins provided the first mechanistic clue to how cells keep proteins from folding into the wrong shapes. Si discusses how the new research expands this view, finding a distinct family of chaperones that helps proteins "shapeshift" when biology requires multiple meaningful states, in contrast to the conventional notion that a protein has just a single "correct" form.
While screening the chaperone proteins in fruit flies, the team made another unexpected connection that potentially broadens the study’s relevance.
Funes was the most striking, but not the only chaperone to affect memory. “If you look at the human version of these genes, they have surprisingly been implicated by genome-wide association studies in schizophrenia,” Patton said. “That’s not something we anticipated.”
Patton cautioned that the overlap does not mean schizophrenia is a “disease of chaperones,” but it opens the door to the possibility that the chaperones could be key factors, potentially acting as mediators.
“Ultimately, chaperones may allow the brain to perceive, process, or store information about the outside world,” Si said. “And in diseases where we do not see the world as it is, like schizophrenia or bipolar disorder, we could imagine chaperones playing a role.”
“While it’s an unknown universe, it’s an exciting one, and we’ll see where we end up,” Si added. “What's remarkable is that we’re now thinking about new ways to treat human diseases, and it all started by studying the sea slug, an organism that, compared to us, is relatively simple.”
Additional authors include Yangyang Yi, Ph.D., Raj Burt, Kevin K-S. Ng, Ph.D., Mayur Mukhi, and Peerzada Shariq Shaheen Khaki, Ph.D.
This work was funded by the Enhanced New Staff Start-up Research Grant from the University of Hong Kong, the Seed Fund for Basic Research from the University of Hong Kong, the Research Grant Council of Hong Kong (award: 17118624), and with institutional support from the Stowers Institute for Medical Research.
Beyond the research: The Inspiration behind a former Stowers Graduate Student's scientific journey
Kyle Patton, Ph.D., a 2024 graduate of the Stowers Graduate School discusses what led him to join the Si Lab and pursue amyloid and memory research. Patton's high school football coach and biology teacher first introduced him to "heat shock proteins" — a family of proteins produced by cells in response to exposure to stressful conditions. Years later he would study them as part of his research at the Stowers Institute.
About the Stowers Institute for Medical Research
Founded in 1994 through the generosity of Jim Stowers, founder of American Century Investments, and his wife, Virginia, the Stowers Institute for Medical Research is a non-profit, biomedical research organization with a focus on foundational research. Its mission is to expand our understanding of the secrets of life and improve life’s quality through innovative approaches to the causes, treatment, and prevention of diseases.
The Institute consists of 20 independent research programs. Of the approximately 500 members, over 370 are scientific staff that include principal investigators, technology center directors, postdoctoral scientists, graduate students, and technical support staff. Learn more about the Institute at www.stowers.org and about its graduate program at www.stowers.org/gradschool.
Media Contact:
Joe Chiodo, Director of Communications
724.462.8529
press@stowers.org
In The News

27 May 2026
From Scientific American, President Alejandro Sánchez Alvarado, Ph.D., shares expert insight on a fascinating new regeneration study.
Read Article
In The News

22 May 2026
Former Stowers Graduate School Summer Scholar Isaac Witte, Ph.D., was featured in the Harvard Gazette ahead of his graduation this month.
Read Article
News
19 May 2026
For Stowers Investigator Linheng Li, Ph.D., a new leukemia study builds on a career spent asking how the places stem cells call home can shape health, disease, and future treatments
Read Article
