In The News

07 January 2026
Investigator Kamena Kostova, named ‘Cell Scientist to Watch’
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
News
KANSAS CITY, MO—Our bodies are perfectly capable of renewing billions of cells every day but fail miserably when it comes to replacing damaged organs such as kidneys. Using the flatworm Schmidtea mediterranea—famous for its capacity to regrow complete animals from minuscule flecks of tissue—as an eloquent example, researchers at the Stowers Institute for Medical Research demonstrated how our distant evolutionary cousins regenerate their excretory systems from scratch.
In the process, the Stowers team led by Howard Hughes Medical Institute and Stowers investigator Alejandro Sanchéz Alvarado, Ph.D., not only established flatworms as a valuable model system to study tissue maintenance and organ regeneration but also provided new clues about the evolutionary origin of mammalian kidneys. Their study is published in the current issue of the journal Development.
“The past ten years of planaria research have shown that planarians use pretty much the same molecular toolkit for building their bodies as other animals,” says first author Jochen Rink, Ph.D., formerly a postdoctoral researcher in the Sanchéz lab and now a group leader at the Max-Planck-Institute of Molecular Cell Biology and Genetics in Dresden, Germany. “Because of this fundamental similarity between all animals, understanding how a worm assembles its excretory system may very well be relevant to understanding kidney physiology and regeneration in humans,” he adds.
Most animals studied in the lab fall within two main branches on the evolutionary tree of life: mice, rats, and salamanders are part of the vertebrate lineage, while fruit flies and the roundworm Ceanorhabditis elegans belong to the lineage encompassing molting animals. “People have drawn conclusions about the evolution of certain attributes by looking exclusively at these two branches,” says Sanchéz Alvarado. “That’s akin to trying to work out your family tree but ignoring your entire maternal lineage.”
In contrast, flatworms, also known as planaria, are part of a branch that has been largely ignored by modern molecular biology. “The so called lophotrochozoa are an incredibly large and diverse group of animals we know very little about,” explains Sanchéz Alvarado.
What was known is that—unlike fruit flies and C. elegans—planaria possess very complex excretory systems akin in many respects to mammalian kidneys. “Planarian protonephridia are so far the only invertebrate model system that combines pressure filtration with filtrate modification similar to mammalian nephrons, the basic functional unit of the kidney,” explains Rink. Their anatomy, however, was not well understood.
When Rink and co-first author Hanh Thi-Kim Vu, Ph.D., a graduate student in the Sanchez Alvarado lab, analyzed their structure with the help of electron microscopy, the scientists found that planarian protonephridia are complex epithelial organs that organized in a consistent and hierarchical manner: a distal tubule branching out into proximal tubules, each of which is topped by a ciliated flame cell—so named because under a microscope the beating of the cilia resembles a flickering candle.
But more importantly, they noticed that the endothelia forming the tubules are composed of a fixed succession of different cell types along their length reminiscent of the mammalian condition. “This suggests to us that the mammalian kidney originated before the branches split,” says Sanchéz Alvarado. “The homologous structures in fruit flies and C. elegans diversified and diverged till they no longer resembled their common ancestor.”
Protonephridia are distributed throughout a flatworm’s body. To study their development researchers could simply cut the animals’ heads off and watch how they regrew the missing body part including excretory tubules within a week. They found that protonephridial tubules originated from a precursor structure, which undergoes extensive branching morphogenesis, the same process that also shapes vertebrate organs such as lung, kidneys or mammary glands.
When Rink interfered with the expression of EGFR5, short for epidermal growth factor receptor, during the regeneration process protonephridia could no longer undergo branching morphogenesis. If he did the same in non-regenerating planaria, they were unable to maintain the structural integrity of their protonephridia, which told him that under normal conditions the organ is constantly maintained through cell renewal.
“We take it for granted that we go to bed with two sets of fully functional kidneys and that we wake up with them the next morning but we don’t understand the fundamental processes that give rise to this very well choreographed maintenance of an organism’s form and function,” says Sanchéz Alvarado. “We can now start to use planaria as a model to begin to understand how an adult animals maintain their form and function over a very long time.”
The work was funded in part by the Howard Hughes Medical Institute and by the National Institutes of Health, National Institute of General Medical Sciences.
About the Stowers Institute for Medical Research
The Stowers Institute for Medical Research is a non-profit, basic biomedical research organization dedicated to improving human health by studying the fundamental processes of life. Jim Stowers, founder of American Century Investments, and his wife Virginia opened the Institute in 2000. Since then, the Institute has spent over a half billion dollars in pursuit of its mission.
Currently the Institute is home to nearly 500 researchers and support personnel; over 20 independent research programs; and more than a dozen technology development and core facilities. Learn more about the Institute at http://www.stowers.org.
In The News

07 January 2026
From the Journal of Cell Science, Investigator Kamena Kostova named a 'Cell Scientist to Watch'
Read Article
#Stowers25: Celebrating 25 Years
06 January 2026
Alejandro Sánchez Alvarado, Ph.D., reflects on a year of discovery, gratitude, and the community that helps support our mission.
Read Article
In The News
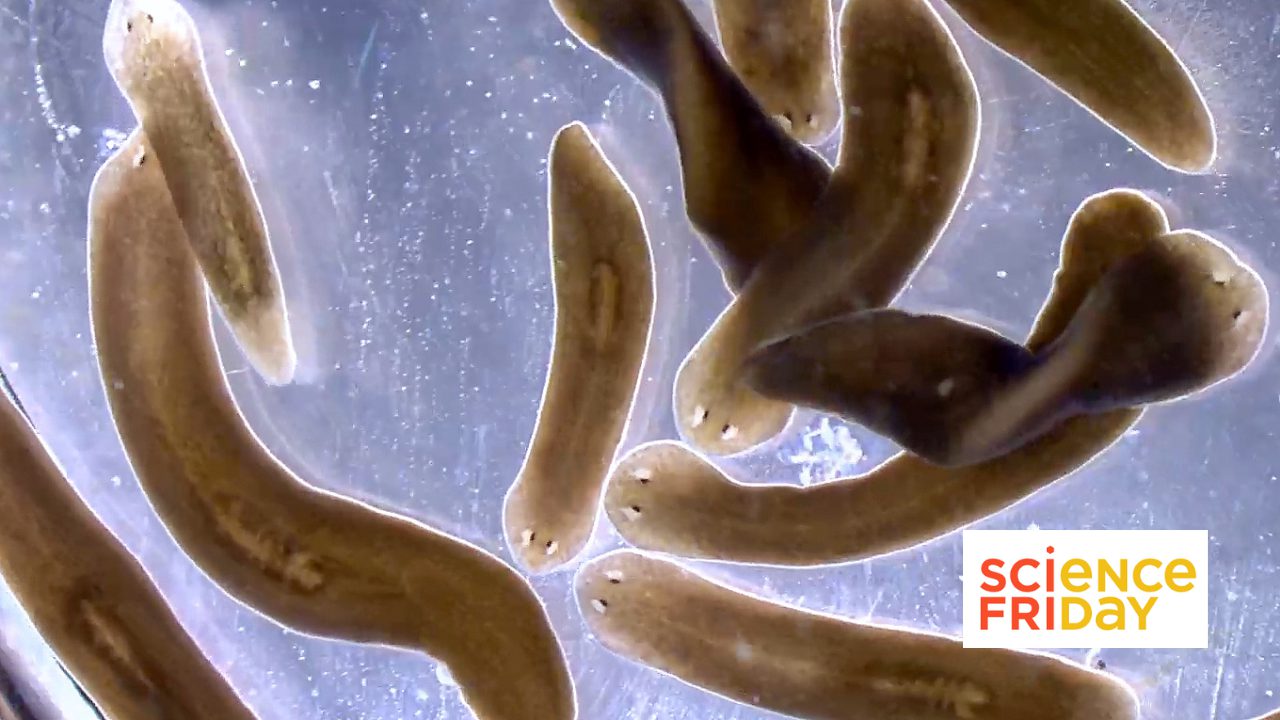
01 January 2026
From Science Friday, President and CSO Alejandro Sánchez Alvarado talks about the science of regeneration and the biology lessons we can carry into the new year.
Read Article
